 
A deep understanding of chemistry is essential to anyone interested in modern biological sciences or medicine, so I really encourage you to take the time to work though all of the chemistry material. 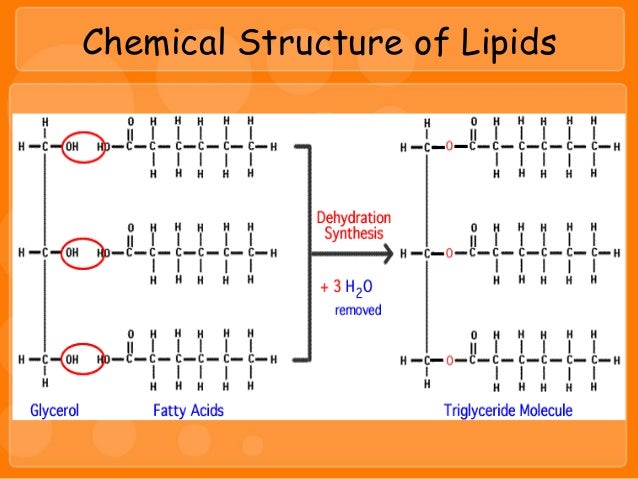
The structure of the fatty acids determines whether or. Fats have glycerol in addition to three fatty acids. Waxes, steroids, phospholipids, and fats are the most common types of lipid groups. Examples of such solvents include acetone and ether. That may look like a lot of work, but you've probably watched many of the videos already under "Chemistry of life". Lipids, as a class of compounds, are insoluble in water but are soluble in other organic solvents. On this page, we’ll learn about the structures of these three types of lipids, as well as their functions in the. There are three main types of lipids: triglycerides, phospholipids, and sterols. If you are not familiar with reduction and oxidation states, then I encourage you to start working through the Chemistry material on KhanAcademy: Lipids are a family of organic compounds that are mostly insoluble in water, meaning they do not mix well with water. How many moles of carbon are present in a gram of glucose (a "typical" carbohydrate)?ĭo the answers to those two sets of questions help you answer your question? How many moles of carbon are present in a gram of tetradecane - a 14 carbon alkane (a reasonable comparison for the tails of the fatty acids found in food)? How would you expect this to affect a oxidative process like cellular respiration? How does this compare with the oxidation state of the carbon in carbohydrates (the other group of macromolecules that are often used to store energy)? What is the oxidation state of the carbons in the fatty acid tail? Solid lipid nanoparticles (SLN) are nanocarriers in the 101000 nm range of a solid core, containing both hydrophilic and hydrophobic active pharmaceutical ingredients. Therefore, I'm going to ask you some questions in response to help you figure out (some of) the answers yourself. This is a good question, but one that I think you have enough information to answer on your own. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
